
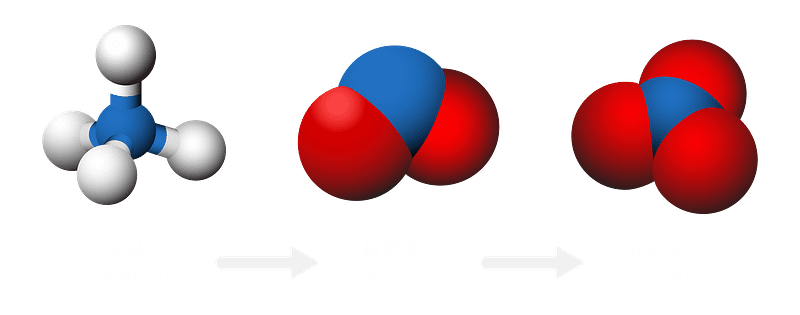



New EPA rules have mandated operators to remove ammonia from wastewater lagoons. Learn why BOD removal, pH, water temp levels and mixing are important.
There are several sources of lagoon ammonia in wastewater lagoons. Learn how lagoons remove ammonia through the process of nitrification.
Excess lagoon sludge impacts the quality of treatment by releasing BOD and ammonia back into the water column. Our video blog explains it all.
Learn how small-footprint NitrOx works within existing infrastructure to cost-effectively meet low ammonia limits year round, no matter how cold it gets.

Whether you’re looking for an ammonia solution for your lagoon or just have a quick question about algae, we’re here for you.
We want to be your #1 resource for all things wastewater lagoons. If we can help you, please don’t hesitate to get in touch.
"*" indicates required fields
We pledge to provide all our customers with the most authentic, long-term solution available. These solutions are built on the shoulders of more than 30 years of professional wastewater engineering experience. Our commitments to excellence, sound research, adaptability, and environmental consciousness separate Triplepoint from our competition.
We stand behind our products and processes. Every installation is backed by our Technology Performance Guarantee: We guarantee that our equipment will do what we say it will do.

800-654-9307
info@lagoons.com
6586 S. Kenton Street
Centennial, CO 80111
Find Us Here