Wastewater lagoon pH is a common testing parameter that is easily taken for granted. Dig a little deeper, however, and pH can give you valuable clues about what’s going on in your lagoon and help you troubleshoot the source of effluent problems.
In this article we’ll talk about what pH is and what various pH readings can reveal about the health of your wastewater lagoon.
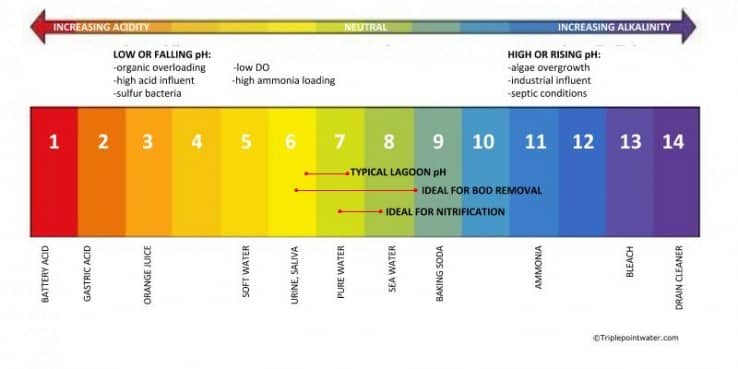
What is pH?

What affects lagoon pH?
Algal activity is responsible for most of the variation in the lagoon. Algae consume CO2 as part of the photosynthesis process, driving up pH. pH is highest in the late afternoon—up to about 7.6—when algae is most active, and lowest just before dawn—at around 6.8—as photosynthesis stops overnight. And because algae thrive in the surface layers of the lagoon, pH tends to be higher near the surface and drop as you get deeper. BOD-removing wastewater lagoon microbes prefer their environment to be on the base to alkaline side, or a pH between 6.5 and 9; a pH outside that range can inhibit aerobic and anaerobic treatment and lead to sludge buildup.
While pH levels in a lagoon can vary by depth or over the course of the day, the pH of the influent is critically important. High or low pH in the influent can indicate septic conditions or industrial discharge. Industrial wastes—like that from wine or dairy processing, which is highly acidic; or which includes metals or cleaning chemicals—can disrupt the biological activity in the lagoon and inhibit treatment. Some lagoons have naturally low alkaline or “soft” water coming in. It’s important for the lagoon operator to be mindful of influent pH levels to be able to identify and correct any abnormal conditions before they affect the balance of the lagoon.
Interestingly, a higher wastewater lagoon pH—towards the alkaline side—has some benefits. Above 8.5, the hydrogen sulfide molecule pulls apart, transforming from rotten-egg smelling H2S into benign hydrogen and hydrosulfide. Lagoon ammonia removal is also pH sensitive: ammonia treatment rates decline significantly at a pH of 6.8 or lower, but get a boost at a pH of 7.5 or higher. In addition, a higher pH controls pathogens and assists in the removal of metals and phosphorus. On the other hand, lagoons with a lower pH, also known as low alkaline or soft water, will be less able to control odors, destroy pathogens, and nitrify. A low or decreasing pH is often caused by low dissolved oxygen (DO) or organic overloading.
Algal activity is responsible for most of the variation in the lagoon; in fact, high effluent pH is almost always attributed to algae. It’s up to the lagoon operator to balance the benefits of a slightly higher pH—odor control and nitrification—with the downside—high TSS and BOD, and sludge accumulation.
How to Adjust Wastewater Lagoon pH
Very high or low pH is often the result of industrial influent, such as acidic dairy wastewater or alkaline cleaning chemicals, or even naturally hard or soft local water, so it’s important for the operator to know what’s coming it and make pH adjustments as necessary to the influent before it enters the lagoon.
In his operations manual, Wastewater Lagoon Troubleshooting, lagoon consultant Steve Harris recommends recirculating water high in DO from the surface of a downstream pond to the surface of a problem pond. Some operators use biological or chemical additives (such as lime) to adjust pH.
A low or decreasing pH due to organic overloading and insufficient DO can be corrected by adding aeration and mixing. Read our blog Seven Signs of an Overloaded Lagoon for more about organic overloading. Mixing, in addition to promoting efficient oxygen transfer, also serves to balance out areas of high or low pH caused by thermal stratification.
Learn how Ares Aeration® can prevent high effluent pH

Controlling algae will prevent most causes of high effluent pH. A properly aerated lagoon should only experience minimal algae growth around its edges during the hottest summer months. The natural habitat for algae is at the surface of the water: If you disrupt that habitat, you also disrupt the algae growth in your wastewater lagoon. The more turbulence in the water, the better your chances are of treating lagoon algae and maintaining optimal pH levels.
Ares Lagoon Aeration can help mitigate lagoon algae and high effluent pH by boosting treatment while increasing mixing and turbulence. Each high flexibility, low maintenance, portable unit combines the mixing advantages of lagoon coarse bubble diffusers to keep solids in suspension with the efficiency of fine bubble diffusers for superior oxygenation. For more information, download our Ares Aeration Literature or contact us and we will be happy to help you with your lagoon aeration project.